Neural Plasticity explains how the nervous system learns and patterns our postures and movement patterns, as well as our memories and sensitivity to pain.
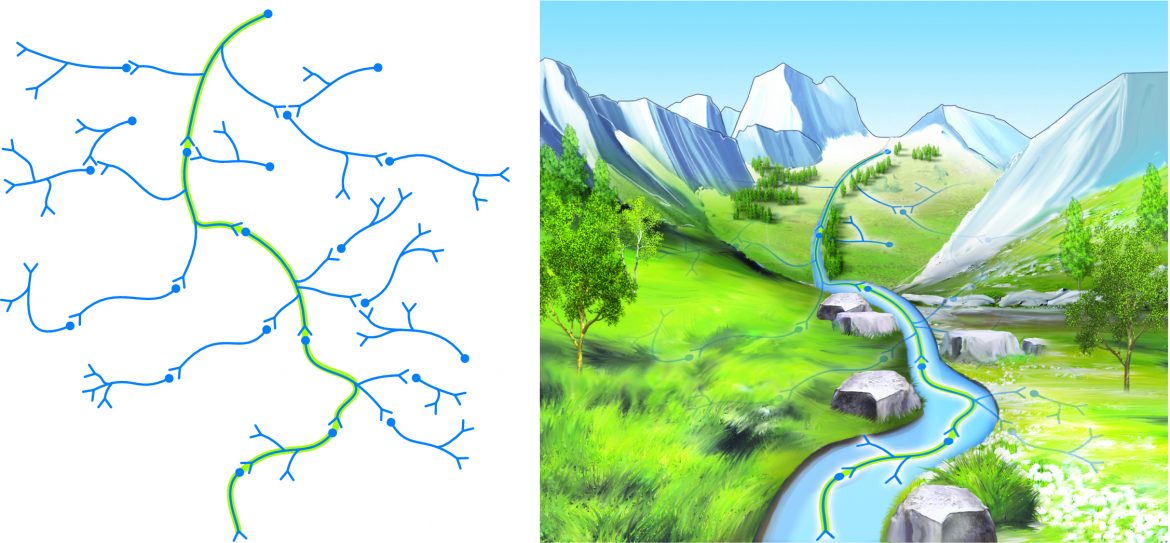
Figure 3. Consolidation of a neural pathway memory pattern is analogous to water etching a deeper and deeper pathway into the side of a mountain over time.
Learning and Memory
The process of learning is the acquisition of knowledge and ability; memory is the retention and storage of knowledge and ability. Memory is stored in the central nervous system by the creation of memory engrams, also called memory patterns, which are, in effect, recordings of what we have learned. Memory storage is usually divided into short-term memory, which lasts for only a few moments; and long-term memory, which can last indefinitely. Whenever any type of long-term memory occurs, whether it is of cognitive intellectual knowledge, a kinesthetic motor skill, emotional feelings, or even pain sensitivity, the concept of neural plasticity is involved. Neural plasticity is the term that describes the brain’s ability to be formed or molded; the term plastic comes from the Greek word plastikos, which relates to molding.
Hence, memory is a recording within the nervous system of our experiences. If the experience is purely intellectual, we form a cognitive memory; if the experience is a physical posture or movement pattern, we form a motor skill memory; if the experience is emotional, we form an emotional memory; and if the experience involves processing sensory pain signals, we can create a memory of pain sensitization. Most memory patterns are a combination of cognitive, motor, sensory, and emotion. In each case, the memory pattern is encoded by the activation/firing of neuronal pathways in a specific sequence.
Note: Review of Nervous System Function
The nervous system is composed of neurons and neuroglial cells (also known simply as glial cells). Neurons are linked together by synaptic connections, and are named for their positions relative to the synapse located between them: the pre-synaptic neuron is located before the synapse; the post-synaptic neuron is located after it (the post-synaptic neuron would then be a pre-synaptic neuron relative to the next synapse along the pathway of neurons). At a synaptic connection, the regions of the membranes where neurotransmitters are released by the pre-synaptic neuron and picked up by the post-synaptic neuron are called active zones. Glial cells are oligodendrocytes, astrocytes, microglial, and ependymal cells in the central nervous system, and Schwann cells in the peripheral nervous system. Neurons comprise 15% of the cells of the brain; glial cells comprise the other 85%.
When a neuron is sufficiently stimulated to reach action potential, an electrical impulse occurs within the neuron. This impulse travels in one direction, from the dendrites/cell body end of the (pre-synaptic) neuron to the ends of the terminal branches of the neuronal axon located at the synapse with the next (post-synaptic) neuron. Because the electrical impulse cannot cross the synaptic connection, the pre-synaptic neuron releases chemical neurotransmitter molecules that cross the synapse to the post-synaptic neuron. These neurotransmitters can have a facilitory or inhibitory effect; in other words, they can facilitate or inhibit the creation of an action potential and therefore nerve impulse in the post-synaptic neuron. Because many (thousands of) pre-synaptic neurons converge on a post-synaptic neuron, whether the post-synaptic neuron is sufficiently facilitated/stimulated to action potential is dependent upon the summation of all facilitory and inhibitory pre-synaptic neuron’s neurotransmitters. If the post-synaptic neuron is sufficiently stimulated, a nerve impulse will be initiated that can then travel to the axonal end of that neuron and synapse with the next post-synaptic neuron. In this manner, impulses travel throughout the neuronal network. In effect, the presence or absence of impulses translates into binary-code information, just as a computer carries 1s and 0s.
This understanding of nervous system function is called the neuron doctrine because it holds that neurons are principally important for nervous system functioning. Although current research does not contradict our knowledge of neuronal functioning, it is beginning to show us that neurons are not the only important actors on the stage when it comes to impulse transmission. We are finding out that glial cells are far more important than previously thought. Glial cells were once thought to provide support functions and be little more than putty that held the neurons together; in fact, the term glia literally means glue. They are now becoming understood to be integral to the functional ability of neurons to carry their electrical impulses and transmit their chemical synaptic messages.
Creating Memory Patterns
The creation of a memory pattern occurs by encoding the activation/firing of specific pathways of neurons in a specific timed sequence. A simple memory pattern might involve only a few neuronal pathways. A more involved pattern might involve many, many neuronal pathways, with a precise interplay of timing.
For example, the motor skill memory pattern to play a simple song on the piano would involve learning to successively flex the metacarpophalangeal and proximal interphalangeal joints of the fingers in a certain sequence, with a specific timing. If it is a simple song such as Mary Had a Little Lamb, the sequence of fingers would be 3-2-1-2-3-3-3, 2-2-2, 3-5-5, 3-2-1-2-3-3-3, 2-2-3-2-1 (3, middle finger; 2, index finger; 1, thumb; and each comma represents a pause). At the same time, the nervous system would have to simultaneously isometrically contract extensor muscles of the wrist joint and flexor muscles of the elbow joint so that the hand and forearm would be positioned such that the fingers can strike the keys, as well as core muscles to hold the trunk and neck in position, while seated to play. If expression is added to the song, then cognitive and/or emotive (limbic system) pathways would be involved, as well as possible other upper extremity joint motions to allow the pianist to strike the keys harder or softer as desired. A memory pattern for this song would involve all these neuronal pathways to engage musculature spatially throughout the body, as well as temporally in sequence and timing to hit the proper order of notes to play the song. And this is a simple song. Imagine if the piece being played is a Beethoven Sonata!
A similar process occurs with a more complicated motor skill, such as hitting a forehand stoke in tennis. Neuronal pathways that encode for hip, knee, and ankle joint motions to place the feet in the right position; elbow, shoulder, shoulder girdle, and spinal joint motions to bring the racquet back; and then swinging the racquet forward with a concerted coordination of all the upper extremity; and spinal, and lower extremity joints for balance and to rotate the body to swing through the ball; all the time tracking the ball with the eyes, cognitively remembering the usual court position of the opponent to determine in which direction to hit the ball, and experiencing the psychological/emotional feelings that are involved in competition.
Long-Term Memory and Neural Plasticity
Therefore, every experience we encounter in our lives results in the formation of an encoded neuronal memory pattern. When first created, this memory pattern is unstable and located in short-term memory. As such, it can usually be accessed for only a few moments. If we want to place a memory into long-term memory, allowing for a more automatic recollection of the memory, we need to stabilize/consolidate it. The key is repetition. The consolidation of a memory pattern results from the repetitive firing of the neurons within the pathways of the memory pattern. With repetition come the functional and structural changes in the neuronal pathways that are the hallmark of neuronal plasticity. Shorter-term changes are functional; longer-term changes are structural.
(Click here for an article on student tips to improve memory.)
Note: Pain Sensitization
Facilitation of pain pathways is termed central sensitization, or simply sensitization. This concept is of extreme importance in the world of manual and movement therapies. If a client has experienced chronic pain, then even if the physical cause of the pain is removed, the facilitation of the pain pathways can create a sensitization of pain in this pathway so that the degree of their pain will be out of proportion to the degree of the physical damage. In these cases, neural plasticity has created a learned pattern of pain that may last for months, years, or indefinitely. The function of sensitization is debated, but it is likely that it acts to heighten the client’s awareness of pain stimuli in a vulnerable region of the body. Given that pain alerts us to physical damage that is occurring or is likely to occur, increased pain sensitization could be viewed as an effective and vigilant warning system that might prevent further damage. The reality is that the pain is overly vigilant and to the client experiencing sensitization, the chronic pain is not appreciated. Interestingly, the reverse of sensitization is also possible. Suppression of pain, termed habituation, can also occur. This results in a lessened degree of pain relative to physical damage that is present.
Note: Emotional Plasticity
Pathways for emotions and psychology have also been shown to be plastic. Indeed, the entire field of talk-therapy could be viewed as a means of becoming aware of unhealthy memory patterns and learning how to change them. The limbic system of the brain is a group of brain structures that are generally considered to be important for emotions. Neuronal emotional plasticity is one reason why a client might have a difficult time letting go of negative feelings. Given the interplay between the motor and limbic systems, emotional plasticity memory patterns also explain why body armoring occurs and why clients may have emotional releases when certain areas of the body are touched.
(Note: Click here for an article on how thinking fast makes you happier.)
Neuronal Plasticity
With early repetition, as a synapse is repetitively fired, the first neural change is functional strengthening of the synapses. This occurs for two reasons. One is that the conductance of potassium (K+) ions within the pre-synaptic neuron is changed; this causes the action potential to lengthen in time, resulting in more neurotransmitter being released. The second functional change is that the transport of the neurotransmitter within the pre-synaptic neuron is increased, making it more available for release. The sum of these two functional changes is that more neurotransmitter is released by the pre-synaptic neuron and therefore picked up by the post-synaptic neuron, increasing the likelihood that the post-synaptic neuron will fire and continue the impulse down the memory pattern’s neuronal pathway (Figure 1).
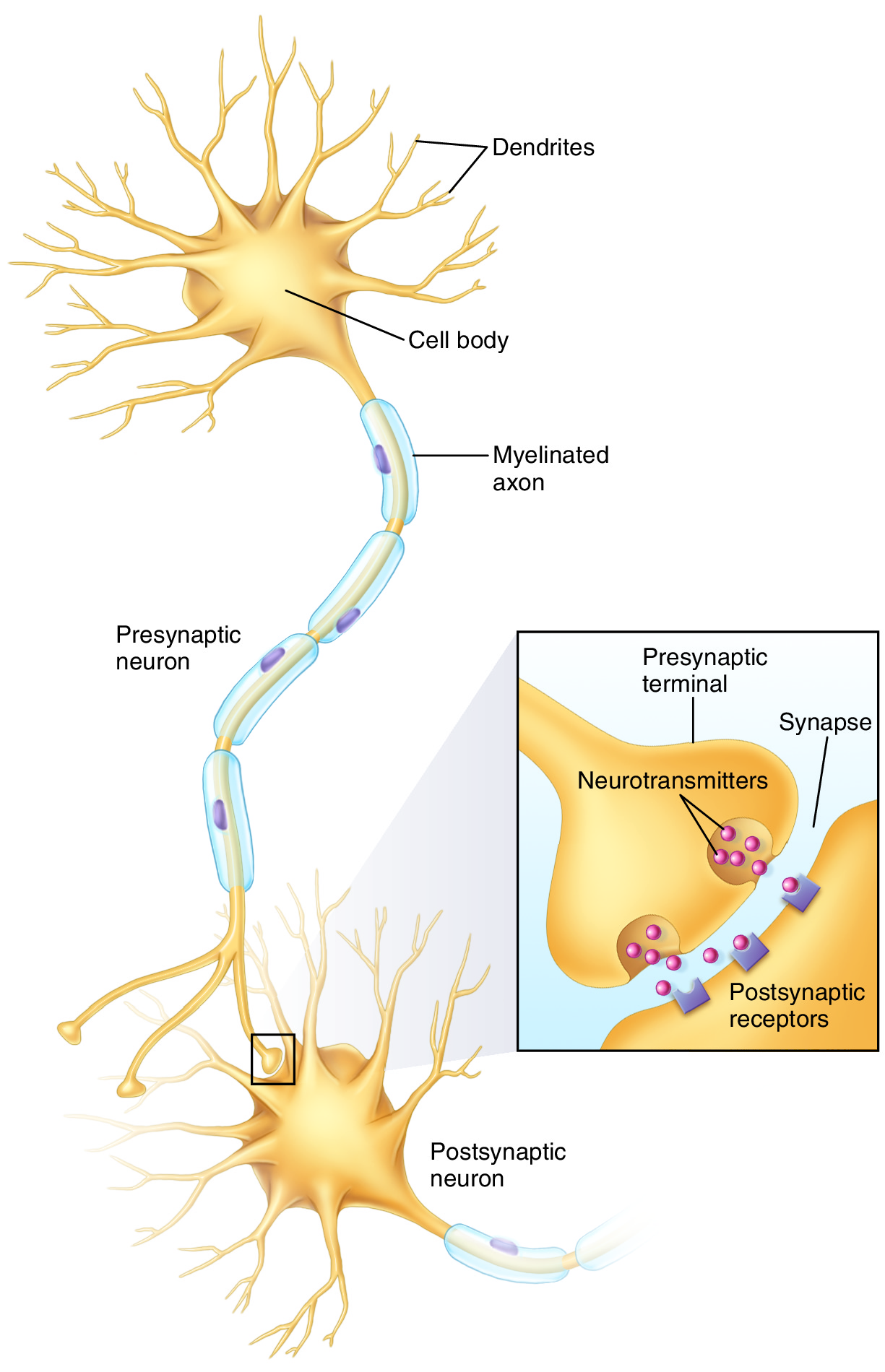
Figure 1. Neurotransmitters are released from the presynaptic neuron, cross the synapse, and are picked up by the postsynaptic neuron. Permission Joseph E. Muscolino. Art work by Giovanni Rimasti.
With continued/later repetition, structural changes begin to occur in both the pre- and post-synaptic neurons. The number of active zones present at the synapses of the pathway increases. In addition, the post-synaptic neuron begins to proliferate its dendritic tree by increasing the number of its dendrites (FIGURE 2). This then allows for the third change, which is that new functional synapses are formed between the pre- and post-synaptic neurons. The result of these structural changes, as with the functional changes, is that more neurotransmitter is picked up by the post-synaptic neuron, increasing the likelihood that it will fire, continuing the impulse down the memory pattern’s neuronal pathway. There is a saying in the world of neurology: “Neurons that fire together wire together.” This describes the concept that when two neurons fire simultaneously, they undergo the functional and structural changes that have been described here as plasticity.
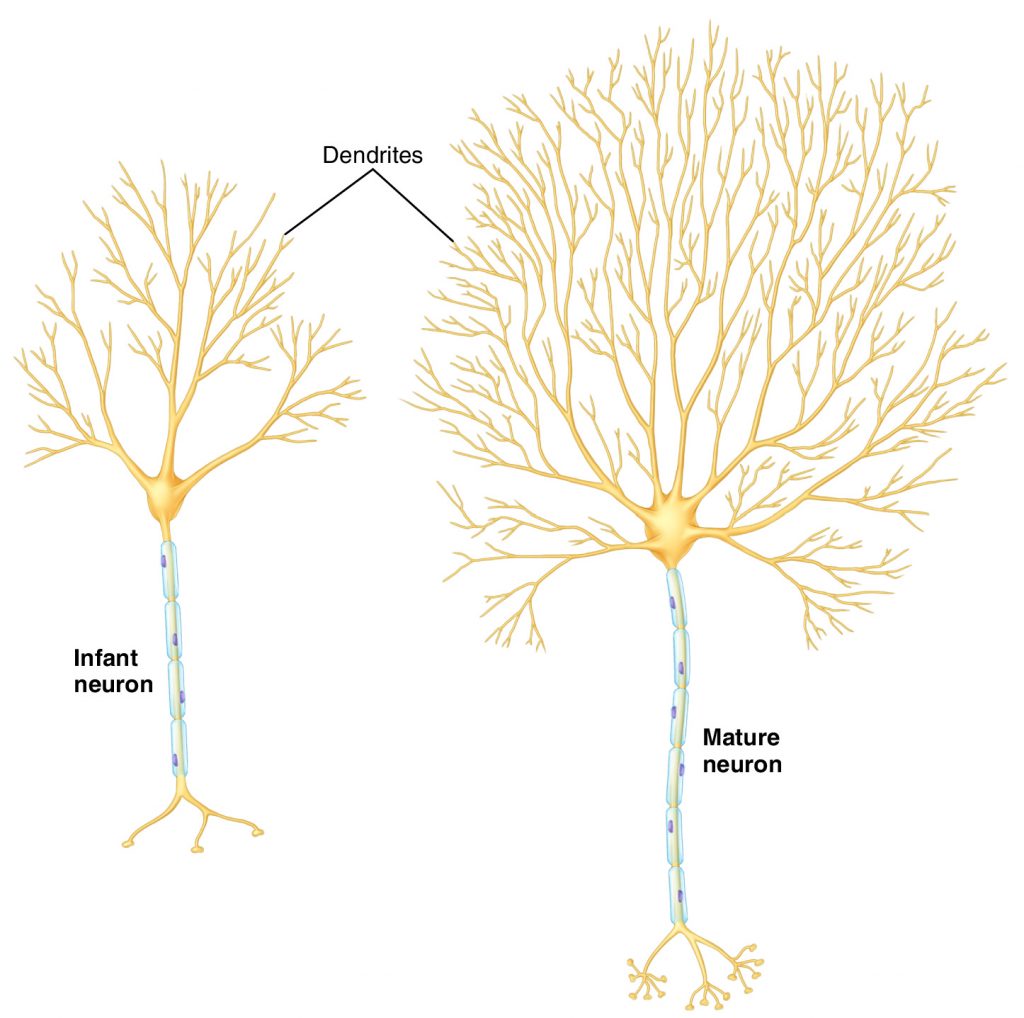
Figure 2. Maturation of a neuron involves proliferation of its dendritic tree. Permission Joseph E. Muscolino. Art work by Giovanni Rimasti.
Neuroglial Plasticity
In addition to neuronal plasticity, neuroglial (glial) cells also exhibit changes with learning and memory. One type of glial plasticity is manifested by oligodendrocytes. Oligodendrocytes function to insulate neuronal axons in the central nervous system by wrapping layers of myelin around them. As a neuron repetitively fires, the oligodendrocyte that myelinates it senses this and responds by increasing the number of myelin layers that ensheath it. This results in greater speed and efficiency of the neuron’s impulse conduction, which can contribute to the efficiency of a learned memory pattern. Astrocytes and microglial cells can also be involved in neural plasticity. These glial cells have been found to contribute to activity in neuronal pain pathways by secreting substances that help to heal the physical damage of the injury. These substances also stimulate the neurons involved, resulting in greater sensitivity to pain. Unfortunately, it has been found that they continue to secrete these substances even after the physical damage is healed. In this manner, glia are implicated in the process of central sensitization to pain.
Note: Brain Mapping Plasticity
As part of the concept of new synaptic connections being formed during the structural phase of neuronal plasticity, the geographic mapping of the cerebral cortex of the brain can also exhibit neural plasticity. This has been shown with both the primary sensory cortex gyrus located in the parietal lobe and the primary motor cortex gyrus located in the frontal lobe. (Because the body areas that these cortical regions represent can be mapped by drawing a small figure of a man, they are termed the sensory homunculus and motor homunculus respectively. The term homunculus literally means little man.) With experience, the body learns and reapportions the neurons of these gyri to the areas of the body that are preferentially used. One study involving the primary sensory cortex showed that after amputation of a digit of the hand, the region of the sensory cortex that had represented that digit was now reapportioned to represent adjacent digits that remained. This concept has also been shown to exist with other sensory regions of the brain. In another study involving the primary motor cortex, if an animal was trained to repeatedly use a specific finger, the region of the motor cortex that represented that finger expanded.
In addition to the plasticity of cortical mapping, reapportioning brain regions also occurs with long-term learning. One of the hallmarks of long-term learning, especially of a motor skill, is that it can be performed in an automatic manner. This occurs by preferentially shifting the command of the motor skill from being seated in the cerebral cortex to being seated primarily in subcortical regions, for example the basal ganglia, situated more deeply in the brain. The advantage to this is that it frees up conscious attention space of the cortex to be devoted to other tasks. This is why a person can perform a motor task in an automatic fashion while thinking about something else entirely. A common example is driving home from work in “auto-drive mode” while lost in thought about the events of the day, only to realize at some point along the way that you have been driving without any conscious attention, and that you have no idea where you are along your route.
Finally, as Charles Leonard, professor of neuroscience of motor control and author, states in The Neuroscience of Human Movement (Mosby, 1998): “Learning is not all in your head.” The spinal cord of the central nervous system can also exhibit neural plasticity. Sensory-motor stretch reflex synaptic connections can also be changed based upon experience.
Learning Through Repetition
The key to neural plasticity is repetition. With each repetition of a cognitive, motor, emotional, or sensory experience, the central nervous system becomes sensitized to that experience and encodes it into a memory pattern via functional and structural changes of neurons and glia. With greater repetition, comes more deeply embedded etching of the pattern. A useful analogy is to compare the neuronal pathway of a memory pattern to water etching a deeper and deeper pathway into the side of a mountain over a period of time (Figure 3). Interestingly, repetition does not have to occur only during our waking hours. When we sleep at night, the brain has been shown to reactivate neuronal pathways of thought and motor patterns that were experienced during the day, thereby strengthening them. It is also during sleep that we move our memory patterns from regions of the cerebral cortex to subcortical regions. This has lead some researchers to posit that the primary function of sleep might actually be to use neural plasticity to prioritize and order the events and experiences of the day into the memory patterns needed for learning.

Figure 3. Consolidation of a neural pathway memory pattern is analogous to water etching a deeper and deeper pathway into the side of a mountain over time. Permission Joseph E. Muscolino. Art work by Giovanni Rimasti.
Application of Neural Plasticity to Manual and Movement Therapy
The concept of neural plasticity is critically important to manual and movement therapy practice. Beyond local effects on myofascial tissues, clinical orthopedic manual and movement therapy is largely geared toward addressing dysfunctional muscle memory patterns located within the nervous system. The difficulty is that these dysfunctional patterns are plastically molded/embedded and become entrenched and difficult to change. This is why the client often experiences a positive temporary effect from the treatment, only to have their symptoms return a short time later. For this reason, it is necessary to create a treatment care and self-care plan for the client that is frequent enough to reverse the embedded dysfunctional pattern. In effect, we are looking to return the client via neural plasticity back to the healthy muscle memory pattern that they once possessed.
(Click here for an article on muscle memory.)
Conclusion
The concept of neural plasticity has been demonstrated in multiple research studies involving cognitive learning, motor learning, and sensory learning. We now understand that far from being a static system, our nervous system is a dynamic plastic system that is constantly forming, reforming, and molding based upon the experiences in our life. Through multifaceted functional and structural changes, neural plasticity integrates these experiences and encodes them into learned memory patterns. We use these memory patterns to understand our world, process pain and other feelings, and to move our bodies in a fluid, graceful, and efficient manner.
Note: This blog post article is modeled from an article originally written in the american massage therapy association‘s massage therapy journal (mtj) in 2011.